
Silver metal reacts with cold dilute nitric acid and produce silver nitrate AgNO 3 salt nitric dioxide NO gas and water as products. Reaction of silver metal and dilute nitric acid Silver reacts with dilute nitric acid and produce silver nitrate AgNO 3 nitrogen dioxide NO 2 and water as products.
Dilute nitric acid on reaction with silver liberatesClass12Subject.
The reaction of silver metal and dilute nitric acid. Silver and cold dilute nitric acid. Silver metal reacts with cold dilute nitric acid and produce silver nitrate AgNO 3 salt nitric dioxide NO gas and water as products. This reaction is a redox reaction oxidation - reduction reaction too.
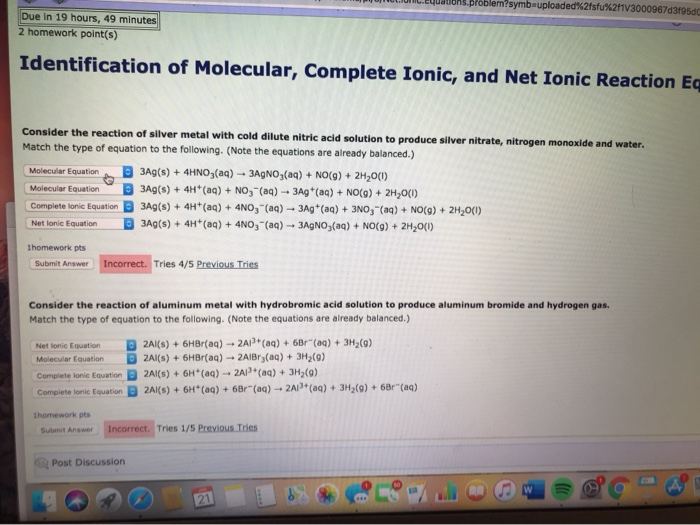
Reaction of silver metal and dilute nitric acid Silver reacts with dilute nitric acid and produce silver nitrate AgNO 3 nitrogen dioxide NO 2 and water as products. Ag HNO 3 AgNO 3 NO 2 H 2 O. Consider the reaction of silver metal with cold dilute nitric acid solution to produce silver nitrate nitrogen monoxide and water.
Match the type of equation to the following. Molecular equation complete ionic equation Net equation or None of the above. Note the equations are already balanced 3Ag s 4H aq 4NO3 aq 3Ag aq.
Silver metal dissolves in hot concentrated sulphuric acid. Silver dissolves in dilute nitric acid HNO 3 8. 3 Ag s 4 HNO 3 aq AgNO 3 aq NO g 2 H 2 O l.
Reaction of silver with acids Silver metal dissolves in hot concentrated sulphuric acid. Silver metal also dissolves in dilute or concentrated nitric acid HNO3. Nitric acid has no action on gold platinum iridium tantalum rhodium and titanium.
All other metals are attacked by the acid often with the formation of nitrates and reduction products of the nitric acid which vary with the temperature and concentration of the acid. Pelouze and Fremy described the reactions of copper and silver with nitric. Silver for example will dissolve in hydrochloric acid or HCl to form silver chloride or AgCl.
Silver chloride however is insoluble in water which means a white solid of AgCl crystals will form in the resulting solution. The reaction of the nitric acid and silver. The Reaction of Metals with Dilute Acid.
Potassium sodium lithium and calcium all react violently with dilute sulfuric acid and dilute hydrochloric acid. It is dangerous to put these metals into an acid. The reaction is similar to the reaction with water forming the metal salt either sulfate or chloride plus H 2g.
Aqueous potassium iodate KIO3 and potassium iodide KI react in the presence of dilute hydrochloric acid as shown belowKIO3aq 5KIaq 6HClaq 3I2aq 6KClaq 3H2OlWhat mass of iodine I2 is formed when 500 mL of 0020 M KIO3 solution reacts with. Silver Calcium carbonate reacts with nitric acid to produce carbon dioxide. A 10 g lump of calcium carbonate was reacted with 20 cm3 of dilute nitric acid.
A The volume of carbon dioxide made in each minute decreases until it remains steady at 83 cm3. Reaction of metals with dilute acids. Metals usually displace hydrogen from dilute acids.
Less reactive metal CuAuAg do not displace hydrogen from dilute acid. Silver and gold do not react with dilute acids. The metals like CuAuAg are less reactive than hydrogenThey do not displace hydrogen from dilute acids.
Dissolution reaction of silver takes place when you place AgAg alloy in Nitric Acid. It depends on Nitric Acid Concentration. 3Ags4HNO3aq —- 3AgNO3lNOg2H2O l -Eqn noI valid for.
Heat the silver nitrate higher temps generate a more vigorous reaction and add a solution of sulfamic acid and water until it stops generating gas. The silver will cement out and stay out of solution much cleaner. Dilution of the solution also helps.
Dilute nitric acid on reaction with silver liberatesClass12Subject. D-BLOCK ELEMENTSBookVK JAISWALBoardIIT JEEYou can ask any doubt fro. You have seen that in the reactions between metals and dilute acids the metal is oxidised to metal ions by the hydrogen ions in the acid and the hydrogen ions are reduced to hydrogen gas.
The problem with nitric acid HNO 3 is that a nitrate ion NO 3 - is a much stronger oxidising agent than a hydrogen ion. Concentrated nitric acid is a very strong oxidator capable of reacting with the noble metals such as copper or silver not gold however. Aluminum and zinc do not react with concentrated nitric acid since a thick hard to dissolve oxidation layer builds passive oxidation that.
The reactions of metals with dilute acids Reactive metals react with dilute acids except dilute nitric acid to produce salts and hydrogen gas. The kind of salts produced depends on the dilute acid used. For example if dilute hydrochloric acid HCl is used a salt called chloride will be produced.
Explain why in a test for halide ions the sample is acidified with dilute nitric acid first. Carbonate ions also produce a white precipitate with silver nitrate solution. The acid reacts with any carbonate ions present.
This removes them so stopping them giving an. Reaction of zinc with dilute sulphuric acid. Zinc sulphate and hydrogen gas are formed when zinc reacts with dilute sulphuric acid.
This method is used in laboratory to produce hydrogen gas. Zn H2SO4 ZnSO4 H2. CopperCopper gold and silver are known as noble metals.
The strong acid HNO3 and strong base react to produce a salt NaNO3 and water H2O. How does sodium react with nitric acid. A metal reacts with an acid to produce a salt and hydrogen gas.
Here sodium reacts with dilute nitric acid to give sodium nitrate salt and hydrogen gas. The method is particularly useful for copper aluminium chromium cobalt indium iron nickel silver and thallium nitrate production. A fast and efficient industrial scale process for producing.
Dilute nitric acid behaves as a typical acid in its reaction with most metals. Magnesium manganese and zinc liberate H 2. Mg 2 HNO 3 MgNO 3 2 H 2 Magnesium nitrate Mn 2 HNO 3 MnNO 3 2 H 2 ManganeseII nitrate Nitric acid can oxidize non-active metals such as copper and silver.
With these non-active or less electropositive metals the products depend on temperature and the acid. Some metals reacts with dilute Nitric acid HNO 3 and produces salt and any of the nitrogen oxides N 2 O NO NO 2 and water. It is because HNO 3 is a strong oxidising agent.
It oxidises the H 2 produced to water and itself gets reduced to any of the nitrogen oxides N 2 O NO NO 2. Cu 4HNO 3 CuNO 3 2 2NO 2 2H 2 O.