Introduction and Purpose Until 1874 commercial salicylic acid was synthesized entirely from natural wintergreen oil. Oct 1 2020 Submission date.
Add 20 mL of methanol to the test tube and swirl until the solid dissolves.
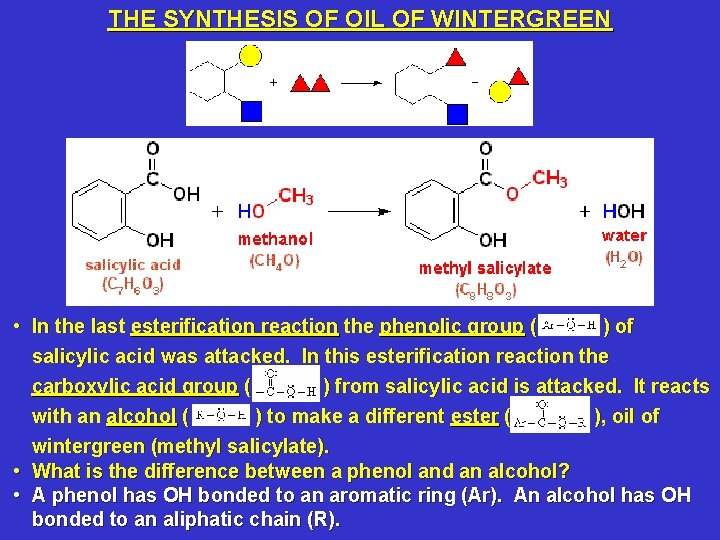
Synthesis of salicylic acid from wintergreen oil. Synthesis of Salicylic Acid from Wintergreen Oil Purpose. The purpose of this lab was to get students familiar with glass materials often used by organic chemists and how to reflux a mixture and to filter its precipitate through teaching students how to create salicylic acid from methyl salicylate wintergreen oil. During the experiment students learns how to use solubility data.
The two step synthesis encompasses aqueous base hydrolysis of wintergreen oil methyl salicylate MS to salicylic acid SA followed by acetylation to acetylsalicylic acid aspirin ASA. Synthesis of Salicylic Acid from Wintergreen Oil. Until 1874 commercial salicylic acid was synthesized entirely from natural wintergreen oil.
In this experiment the salicylic acid is reproduced using methyl salicylate the major constituent of wintergreen oil. Synthesis of Salicylic Acid from Wintergreen Oil I. Introduction and Purpose Until 1874 commercial salicylic acid was synthesized entirely from natural wintergreen oil.
In this experiment the salicylic acid is reproduced using methyl salicylate the major constituent of wintergreen oil and later compared to salicylic acid made from benzene. Synthesis of Salicylic Acid from Wintergreen Oil Objective Preparation of salicylic acid organic synthesis from methyl salicylate utilizing previously used procedure from the nineteenth century. The final product will then be evaluated in comparison to salicylic acid made from benzene.
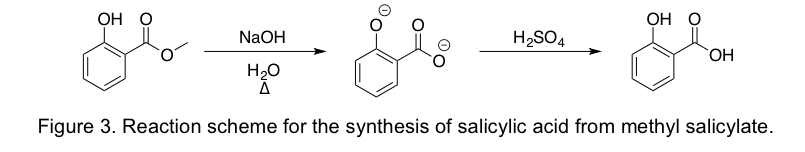
The purpose of this experiment was to perform a base-catalyzed hydrolysis reaction while converting oil of wintergreen to salicylic acid. The full balanced molecular equation for the salicylic acid synthesis is C 8 H 8 O 3 2 NaOH H 2 SO 4 - C 4 H 6 O 3 Na 2 SO 4 CH 3 OH H 2 O. An illustrated equation showing compounds to be isolated or synthesized is shown below.
In this lab salicylic acid was synthesized from wintergreen oil which involves the conversion of functional groups. Changing the functional group of a compound to create a similar compound changes the physical traits of the compound. Synthesis is a basic way.
Reproducibility as regards to yield and purity of the API. The two step synthesis encompasses aqueous base hydrolysis of wintergreen oil methyl salicylate MS to salicylic acid SA followed by acetylation to acetylsalicylic acid aspirin ASA. Synthesis of Aspirin O OH O O O O Na Na OH OH O OH OH O OH O O O O O O Na O O O MW 15215 MF C 8 H 8 O 3.
Experiment 4 Heating Under Reflux Synthesis of Salicylic Acid from Wintergreen Oil. The objective of the experiment is to prepare the salicylic acid from methyl salicylate. Synthesis of Wintergreen Oil 1.
Fill a 400 mL beaker approximately halfway with tap water. Place it on a hot plate. Collect a glass thermometer and split stopper.
Slide the thermometer through the hole. Clamp the thermometer in the water approximately 1 cm. Synthesis of salicylic acid from wintergreen oil - YouTube.
Synthesis of salicylic acid from wintergreen oil. If playback doesnt. Hydrolysis of oil of wintergreen.
2 Synthesis of salicylic acid from oil of wintergreen Methyl Salicylate Salicylic Acid Molecular Mass 15215 g mol-1 13812 g mol-1 Stoichiometry 1 1 Density 117 g mL-1 Solid Moles 003 mol Theoretical. 003 mol Mass 468 g 4 mL Theoretical. 414 g Procedure Add methyl salicylate 4 mL and 6 M sodium.
LAB REPORT Experiment 4 Heating Under Reflux Synthesis of Salicylic Acid from Wintergreen Oil Course. CHEM 222 Experiment date. Oct 1 2020 Submission date.
The objective of the experiment is to prepare the salicylic acid from methyl salicylate. This experiment is performed to see if there is a difference between the. Synthesis of Salicylic Acid from Wintergreen Oil Ethan Gorman Sly.
61615 Abstract The purpose of the experiment was to show that salicylic acid can be synthesized from benzene in the lab. After the synthesis recrystallization is necessary to purify the solid and then measure the melting range of the dry product against the melting points of salicylic acid in the lab manual. Salicylic acid from wintergreen oil methyl salicylate Written and tested with success by Sciencemadness member Alexleyenda.
Wintergreen oil which is at least 98 methyl salicylate can be found for a good price in many drugstores. To remove any left over H2SO4 or other left over starting materials from the Salicylic Acid solvent Prevents loss of product yield What can you conclude if the Melting Point of the Salicylic Acid you just synthesized with Wintergreen Oil is 152-155 and the 11 Mix of your product and synthetic from Benzene salicylic acid is 151-154. Synthesis of Salicylic Acid from Wintergreen Oil Abstract.
The purpose of this experiment is to take methyl salicylate wintergreen oil and by heating it under reflux with NaOH as a solvent and then cooling the mixture with H2SO4 as another solvent synthesize salicylic acidThe final step involves purify the product. Experiment 4 Synthesis of Salioylic Acid from Wintergreen oil Synthesis of Salicylic Acid from Wintergreen Oil Heating Under Reflux Dr Ex Preparation and Purification of Solids Operations OP-2 Using Specialized Glassware OP-7b Smooth Boiling Devices OP-7c Heating Under Reflux OP-4 Weighing OP-7a Heat Sources OP-16 Vacuum Filtration OP-26 Washing. Start studying Experiment 4 Synthesis of Salicylic Acid from Wintergreen oil.
Learn vocabulary terms and more with flashcards games and other study tools. Synthesis of Oil of Wintergreen Pre- Lab Discussion. Methyl salicylate C6H4 HO COOCH3 also known as salicylic acid methyl ester oil of wintergreen is a natural product of many species of plants.
Methyl salicylate may be used by plants as a pheromone to warn other plants of. Conclusion Salicylic acid was able to be synthesized from the naturally occurring wintergreen oil and appeared to exhibit the same melting points as the commercially derived. This concludes that both synthetic salicylic acid from petroleum production and naturally derived salicylic from wintergreen oil are the same compound and commercially derived is able to be produced in a more pure state.
Synthesis of Wintergreen Oil 1. Fill a 400 mL beaker approximately halfway with tap water. Place it on a hot plate.
Collect a glass thermometer and split stopper. Slide the thermometer through the hole. Clamp the thermometer in the water approximately 1 cm.
Synthesis of Aspirin and Oil of Wintergreen Revision F8 Page 4 of 7 Synthesis of Oil of Wintergreen 1. Weigh 025 grams of salicylic acid into a 10-15-cm test tube. This weight does not need to be recorded.
Add 20 mL of methanol to the test tube and swirl until the solid dissolves.